Suppose that we start from a system in thermal equilibrium.
According to statistical mechanics, each of the
![]() ,
say, accessible states are equally likely. Let us now remove, or relax, some of the
constraints imposed on the system. Clearly, all of the microstates formally
accessible to the system are still accessible, but many additional states will,
in general, become accessible. Thus, removing or relaxing constraints can only have
the effect of increasing, or possibly leaving unchanged, the number of
microstates accessible to the system. If the final number of accessible states
is
,
say, accessible states are equally likely. Let us now remove, or relax, some of the
constraints imposed on the system. Clearly, all of the microstates formally
accessible to the system are still accessible, but many additional states will,
in general, become accessible. Thus, removing or relaxing constraints can only have
the effect of increasing, or possibly leaving unchanged, the number of
microstates accessible to the system. If the final number of accessible states
is
![]() , then we can write
, then we can write
| (211) |
As a simple example, consider a system
consisting of a box divided into two regions of equal volume. Suppose that,
initially, one region is filled with gas and the other is empty. The constraint
imposed on the system is, thus, that the coordinates of all of the molecules must
lie within the filled region. In other words, the volume accessible to the
system is ![]() , where
, where ![]() is half the volume of the box. The constraints
imposed on the system can be relaxed by removing the partition and allowing gas to
flow into both regions. The volume accessible to the gas is now
is half the volume of the box. The constraints
imposed on the system can be relaxed by removing the partition and allowing gas to
flow into both regions. The volume accessible to the gas is now ![]() .
Immediately after the partition is removed, the system is
in an extremely improbable state.
We know, from Sect. 3.8, that at constant energy the variation of the number
of accessible states of an ideal gas with the volume is
.
Immediately after the partition is removed, the system is
in an extremely improbable state.
We know, from Sect. 3.8, that at constant energy the variation of the number
of accessible states of an ideal gas with the volume is
| (213) |
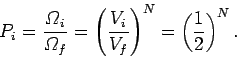 |
(214) |
| (215) |
This discussion can also be phrased in terms of
the parameters
![]() of the system.
Suppose that a constraint is removed.
For instance, one of the parameters,
of the system.
Suppose that a constraint is removed.
For instance, one of the parameters, ![]() , say, which originally had the value
, say, which originally had the value
![]() , is now allowed to vary. According to statistical mechanics, all states
accessible to the system are equally likely. So, the probability
, is now allowed to vary. According to statistical mechanics, all states
accessible to the system are equally likely. So, the probability ![]() of finding the
system in equilibrium with the parameter
in the range
of finding the
system in equilibrium with the parameter
in the range ![]() to
to ![]() is just proportional
to the number of microstates in this interval: i.e.,
is just proportional
to the number of microstates in this interval: i.e.,
| (216) |
If some of the constraints of an isolated system are removed then the parameters of the system tend to readjust themselves in such a way that
Suppose that the final equilibrium state has been reached, so that the systems in the
ensemble are uniformly distributed over the
![]() accessible final states.
If the original constraints are reimposed then the systems
in the ensemble still
occupy these
accessible final states.
If the original constraints are reimposed then the systems
in the ensemble still
occupy these
![]() states with equal probability. Thus, if
states with equal probability. Thus, if
![]() , simply restoring the constraints does not restore the initial situation.
Once the systems are randomly distributed over the
, simply restoring the constraints does not restore the initial situation.
Once the systems are randomly distributed over the
![]() states they cannot
be expected to spontaneously move out of some of these states and occupy a
more restricted class of states merely in
response to the reimposition of a constraint.
The initial condition can also not be restored by removing further constraints. This
could only lead to even more states becoming accessible to the system.
states they cannot
be expected to spontaneously move out of some of these states and occupy a
more restricted class of states merely in
response to the reimposition of a constraint.
The initial condition can also not be restored by removing further constraints. This
could only lead to even more states becoming accessible to the system.
Suppose that some process occurs in which an isolated system
goes from some initial configuration to some final configuration. If the
final configuration is such that the imposition or removal of constraints
cannot by itself restore the initial condition then
the process is deemed irreversible. On the other hand, if it is such that the
imposition or removal of constraints can restore the initial condition
then the
process is deemed reversible. From what we have already said, an irreversible
process is clearly one in which the removal of constraints leads to a situation
where
![]() . A reversible process corresponds to the special
case where the removal of constraints does not change the number of accessible
states, so that
. A reversible process corresponds to the special
case where the removal of constraints does not change the number of accessible
states, so that
![]() . In this situation, the systems
remain
distributed with equal probability over these states irrespective of whether the
constraints are imposed or not.
. In this situation, the systems
remain
distributed with equal probability over these states irrespective of whether the
constraints are imposed or not.
Our microscopic definition of irreversibility is in accordance with the macroscopic definition discussed in Sect. 3.6. Recall that on a macroscopic level an irreversible process is one which ``looks unphysical'' when viewed in reverse. On a microscopic level it is clearly plausible that a system should spontaneously evolve from an improbable to a probable configuration in response to the relaxation of some constraint. However, it is quite clearly implausible that a system should ever spontaneously evolve from a probable to an improbable configuration. Let us consider our example again. If a gas is initially restricted to one half of a box, via a partition, then the flow of gas from one side of the box to the other when the partition is removed is an irreversible process. This process is irreversible on a microscopic level because the initial configuration cannot be recovered by simply replacing the partition. It is irreversible on a macroscopic level because it is obviously unphysical for the molecules of a gas to spontaneously distribute themselves in such a manner that they only occupy half of the available volume.
It is actually possible to quantify irreversibility.
In other words, in addition to
stating that
a given process is irreversible, we can also give some indication
of how irreversible it is. The parameter which measures irreversibility is
just the number of accessible states ![]() .
Thus, if
.
Thus, if ![]() for an isolated
system spontaneously
increases then the process is irreversible, the degree of irreversibility
being proportional to the amount of
the increase. If
for an isolated
system spontaneously
increases then the process is irreversible, the degree of irreversibility
being proportional to the amount of
the increase. If ![]() stays the same then the process
is reversible. Of course, it is unphysical for
stays the same then the process
is reversible. Of course, it is unphysical for ![]() to ever spontaneously
decrease. In symbols, we can write
to ever spontaneously
decrease. In symbols, we can write
| (219) |
| (221) |
| (222) |
| (223) |
| (224) |
| (225) |
| (226) |
One way of thinking of the number of accessible states ![]() is that it is a measure
of the disorder associated with a macrostate. For a system exhibiting
a high degree of order we would expect a strong correlation between the motions
of the individual particles. For instance, in a fluid there might be a strong tendency
for the particles to move in one particular direction, giving rise to
an ordered flow of the
system in that direction.
On the other hand, for a system exhibiting a low degree of order we expect
far less correlation between the motions of individual particles. It follows that,
all other things being equal, an ordered system is more constrained than a disordered
system, since the former is excluded from microstates in which there is not
a strong correlation between individual particle motions, whereas the latter is not.
Another way of saying this is that an ordered system has less accessible microstates
than a corresponding disordered system. Thus, entropy is
effectively a measure of the disorder
in a system (the disorder increases with
is that it is a measure
of the disorder associated with a macrostate. For a system exhibiting
a high degree of order we would expect a strong correlation between the motions
of the individual particles. For instance, in a fluid there might be a strong tendency
for the particles to move in one particular direction, giving rise to
an ordered flow of the
system in that direction.
On the other hand, for a system exhibiting a low degree of order we expect
far less correlation between the motions of individual particles. It follows that,
all other things being equal, an ordered system is more constrained than a disordered
system, since the former is excluded from microstates in which there is not
a strong correlation between individual particle motions, whereas the latter is not.
Another way of saying this is that an ordered system has less accessible microstates
than a corresponding disordered system. Thus, entropy is
effectively a measure of the disorder
in a system (the disorder increases with ![]() ).
With this interpretation, the second law of thermodynamics reduces to the statement
that isolated systems tend to become more disordered with time, and can never
become more ordered.
).
With this interpretation, the second law of thermodynamics reduces to the statement
that isolated systems tend to become more disordered with time, and can never
become more ordered.
Note that the second law of thermodynamics only applies to isolated systems. The entropy of a non-isolated system can decrease. For instance, if a gas expands (at constant energy) to twice its initial volume after the removal of a partition, we can subsequently recompress the gas to its original volume. The energy of the gas will increase because of the work done on it during compression, but if we absorb some heat from the gas then we can restore it to its initial state. Clearly, in restoring the gas to its original state, we have restored its original entropy. This appears to violate the second law of thermodynamics because the entropy should have increased in what is obviously an irreversible process (just try to make a gas spontaneously occupy half of its original volume!). However, if we consider a new system consisting of the gas plus the compression and heat absorption machinery, then it is still true that the entropy of this system (which is assumed to be isolated) must increase in time. Thus, the entropy of the gas is only kept the same at the expense of increasing the entropy of the rest of the system, and the total entropy is increased. If we consider the system of everything in the Universe, which is certainly an isolated system since there is nothing outside it with which it could interact, then the second law of thermodynamics becomes:
The disorder of the Universe tends to increase with time and can never decrease.
An irreversible process is clearly one which increases the disorder of the Universe, whereas a reversible process neither increases nor decreases disorder. This definition is in accordance with our previous definition of an irreversible process as one which ``does not look right'' when viewed backwards. One easy way of viewing macroscopic events in reverse is to film them, and then play the film backwards through a projector. There is a famous passage in the novel ``Slaughterhouse 5,'' by Kurt Vonnegut, in which the hero, Billy Pilgrim, views a propaganda film of an American World War II bombing raid on a German city in reverse. This is what the film appeared to show:
``American planes, full of holes and wounded men and corpses took off backwards from an airfield in England. Over France, a few German fighter planes flew at them backwards, sucked bullets and shell fragments from some of the planes and crewmen. They did the same for wrecked American bombers on the ground, and those planes flew up backwards and joined the formation.
The formation flew backwards over a German city that was in flames. The bombers opened their bomb bay doors, exerted a miraculous magnetism which shrunk the fires, gathered them into cylindrical steel containers, and lifted the containers into the bellies of the planes. The containers were stored neatly in racks. The Germans had miraculous devices of their own, which were long steel tubes. They used them to suck more fragments from the crewmen and planes. But there were still a few wounded Americans, though, and some of the bombers were in bad repair. Over France, though, German fighters came up again, made everything and everybody as good as new.''Vonnegut's point, I suppose, is that the morality of actions is inverted when you view them in reverse.
What is there about this passage which strikes us as surreal and fantastic? What is there that immediately tells us that the events shown in the film could never happen in reality? It is not so much that the planes appear to fly backwards and the bombs appear to fall upwards. After all, given a little ingenuity and a sufficiently good pilot, it is probably possible to fly a plane backwards. Likewise, if we were to throw a bomb up in the air with just the right velocity we could, in principle, fix it so that the velocity of the bomb matched that of a passing bomber when their paths intersected. Certainly, if you had never seen a plane before it would not be obvious which way around it was supposed to fly. However, certain events are depicted in the film, ``miraculous'' events in Vonnegut's words, which would immediately strike us as the wrong way around even if we had never seen them before. For instance, the film might show thousands of white hot bits of shrapnel approach each other from all directions at great velocity, compressing an explosive gas in the process, which slows them down such that when they meet they fit together exactly to form a metal cylinder enclosing the gases and moving upwards at great velocity. What strikes us as completely implausible about this event is the spontaneous transition from the disordered motion of the gases and metal fragments to the ordered upward motion of the bomb.