


Next: Photon Polarization
Up: Fundamental Concepts
Previous: Fundamental Concepts
The necessity for a departure from
classical physics at the microscopic (i.e., atomic, molecular, and particle) level is amply demonstrated by the following well-known phenomena:
- Anomalous Atomic and Molecular Stability:
- According to classical
physics, an electron orbiting an atomic nucleus undergoes acceleration and should, therefore, lose energy via the continuous emission
of electromagnetic radiation [49], causing it to gradually spiral in towards the nucleus. (See Exercise 1.) Experimentally,
this is not observed to happen.
- Anomalously Low Atomic and Molecular Specific Heats:
- According to
the equipartition theorem of classical statistical thermodynamics, each degree of freedom of
an atom or molecule should contribute
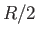 to the molar specific heat capacity of a macroscopic system made up of
a great many such atoms or molecules, where
to the molar specific heat capacity of a macroscopic system made up of
a great many such atoms or molecules, where 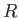 is the molar ideal gas constant [91].
In fact, only the translational, and some rotational, degrees of freedom seem
to contribute. The vibrational degrees of freedom appear to make no contribution
at all
(except at very high temperatures) [91]. Incidentally, this fundamental
problem with classical physics was known and appreciated by the middle of the
nineteenth century. Stories that physicists at the commencement of the twentieth century thought that
classical physics explained everything, and that there was nothing left to
discover, are largely apocryphal [46].
is the molar ideal gas constant [91].
In fact, only the translational, and some rotational, degrees of freedom seem
to contribute. The vibrational degrees of freedom appear to make no contribution
at all
(except at very high temperatures) [91]. Incidentally, this fundamental
problem with classical physics was known and appreciated by the middle of the
nineteenth century. Stories that physicists at the commencement of the twentieth century thought that
classical physics explained everything, and that there was nothing left to
discover, are largely apocryphal [46].
- Ultraviolet Catastrophe:
- According to classical statistical thermodynamics, the equilibrium energy
density of an electromagnetic field contained within a vacuum cavity whose walls are held at a fixed temperature is infinite, due to a divergence of
energy carried by short-wavelength modes. This divergence is called the ultraviolet catastrophe [35]. Experimentally, there is no such
divergence, and the total energy density is finite [91].
- Wave-Particle Duality:
- Classical physics treats waves and
microscopic particles as completely distinct phenomena. However, various experiments (e.g., the photoelectric effect [37,74], Compton scattering [22], and
electron diffraction [25,111]) demonstrate that waves sometimes act as if they
were streams of particles, and streams of particles sometimes act as if they
were waves [26]. This behavior is completely inexplicable within the framework of
classical physics.



Next: Photon Polarization
Up: Fundamental Concepts
Previous: Fundamental Concepts
Richard Fitzpatrick
2016-01-22