Next: One-Dimensional Fourier Optics Up: Wave Optics Previous: Multi-Slit Interference Contents
Suppose that a very thin film of air is trapped between two pieces of glass, as shown in Figure 10.9. If monochromatic light is normally (or almost normally) incident on the film then some of the light is reflected from the interface between the bottom of the upper plate and the air, and some is reflected from the interface between the air and the top of the lower plate. These two reflected light rays interfere either destructively or constructively with one another.
Let 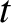 be the thickness of the air film. The difference in distance traveled
between the two light rays shown in the figure is
be the thickness of the air film. The difference in distance traveled
between the two light rays shown in the figure is
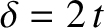 .
This difference introduces a phase difference
.
This difference introduces a phase difference
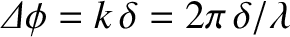 between the rays.
Naively, we might expect that constructive interference would occur when
between the rays.
Naively, we might expect that constructive interference would occur when
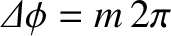 , where
, where 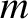 is an integer,
and destructive interference would occur when
is an integer,
and destructive interference would occur when
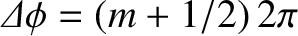 . However, this is not the
case, because an additional phase difference is introduced between
the rays on reflection. The first ray is reflected at an interface
between an optically dense medium (glass) and a less dense medium (air).
There is no phase change on reflection from such an interface. (See Section 6.8.)
The second ray is reflected at an interface
between an optically less dense medium (air) and a dense medium (glass). There is a
. However, this is not the
case, because an additional phase difference is introduced between
the rays on reflection. The first ray is reflected at an interface
between an optically dense medium (glass) and a less dense medium (air).
There is no phase change on reflection from such an interface. (See Section 6.8.)
The second ray is reflected at an interface
between an optically less dense medium (air) and a dense medium (glass). There is a
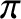 radian phase change on reflection from such an interface. (See Section 6.8.) Thus, an additional
radian phase change on reflection from such an interface. (See Section 6.8.) Thus, an additional 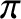 radian phase change
is introduced between the two rays, which is equivalent to an
additional path difference of
radian phase change
is introduced between the two rays, which is equivalent to an
additional path difference of 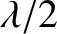 . When this additional
phase change is taken into account, the condition for constructive
interference becomes
. When this additional
phase change is taken into account, the condition for constructive
interference becomes
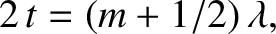 |
(10.45) |
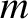 is an integer. Similarly, the condition for
destructive interference becomes
is an integer. Similarly, the condition for
destructive interference becomes
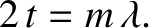 |
(10.46) |
For white light, the previous criteria yield constructive interference for some wavelengths, and destructive interference for others. Thus, the light reflected back from the film exhibits those colors for which constructive interference occurs.
If the thin film consists of water, oil, or some other transparent material,
of refractive index 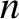 then the results are basically the same as those for
an air film, except that the wavelength of the light in the
film is reduced from
then the results are basically the same as those for
an air film, except that the wavelength of the light in the
film is reduced from 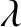 (the vacuum wavelength) to
(the vacuum wavelength) to 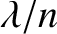 .
It follows that the modified criteria for constructive and destructive
interference are
.
It follows that the modified criteria for constructive and destructive
interference are
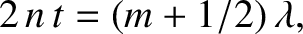 |
(10.47) |
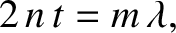 |
(10.48) |